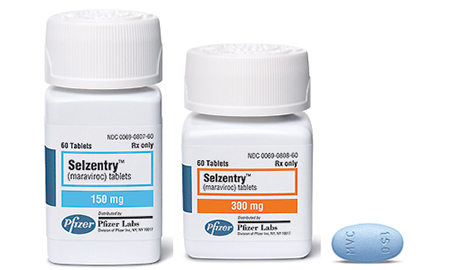
The FDA is encouraging drug makers to incorporate more women and minorities in their clinical trials, and a study in the journal Drug Metabolism and Disposition highlights a key reason why greater diversity in the pool of potential patients is needed. Researchers from Johns Hopkins assert that tests of the HIV medication maraviroc—Pfizer’s Selzentry—relied mostly on European Americans who generally have at least one faulty CYP3A5 gene, resulting in low levels of a protein that removes the drug from the body. This means that drug levels among these patients remained at a given level. The problem: African-Americans generally produce that protein, so the European doses may not be enough because the protein is pushing the drug out of the blood stream at a much higher pace, and this population may need a larger dose.
Medical cost discussions often focus on diseases and the cost of containment, management and potential lost income from illness. A study by researchers indicates that transient conditions also need attention, and menopause was one that grabbed theirs. The Wall Street Journal reports that women who experience hot flashes but do not treat them may be forced to take more time away from the office, or may end up not working at all, compared to those who do not experience the waves of heat (officially called vasomotor symptoms). The cost: 1.5 million more visits to healthcare providers among those hot-flash patients than among those without VMS, which the Journal says comes to close to $340 million in additional office visit payments. Researchers said the lost work hours came to about $27.7 million in lost productivity.
Heroin use is rising in New York, and so are related overdoses. The New York Times reports overdoses killed more city residents last year than in any year since 2003 and that the death toll “has more than doubled over the last three years.” The uptick parallels a prescription painkiller medication crackdown which has made it harder for patients to get medications like oxycodone and hydrocodone. The inverse relationship is not unusual: Florida found a similar pattern which the Centers for Disease Control and Prevention indicated could indicate abusers switched from prescription opioids to more easily accessible ones.
Pharmacists can play a key role in helping patients comply with medication regimens. Drug Store News reports that Shoppers Drug Mart and a Canadian nonprofit interviewed about 500 Canadians who had reviewed their medications with one of the drug chain’s pharmacists and found that 36% said they left understanding potential side effects and 83% said the pharmacist consult made remembering how to make their medication easier.
The National Institute of Allergy and Infectious Diseases—part of the National Institutes of Health—will begin human clinical trials of GlaxoSmithKline’s experimental Ebola vaccine next week. The tests will use uninfected adults to assess if the vaccine triggers a sufficient immune response and are part of a roster of clinical trials that the NIH and partners (including the UK’s Wellcome Trust and Britain’s Medical Research Council) are undertaking to accelerate testing of experimental treatments. The GSK vaccine is based on a chimpanzee cold virus and includes DNA from two Ebola strains. The virus will not replicate and induce the virus, but the hope is it will trigger an immune response. Thursday’s announcement comes just as the World Health Organization issued an estimate that it expects more than 20,000 people will be infected by the Ebola virus before the outbreak is contained. Bloomberg reports that the WHO estimates it will cost around $490 million to contain the virus and that more than 40% of infections have happened within the last 21 days. The virus has torn through Liberia, Guinea, Sierra Leone and Nigeria. Bloomberg also reports that Nigeria has reported its first Ebola death outside of Lagos, in Port Harcourt.