Pfizer took aim at Johnson & Johnson’s Tylenol franchise with a campaign offering free Advil and urging consumers to clean out their medicine cabinets and trash recalled drugs.
The campaign includes TV, print and web ads, along with new websites and messaging savaging Tylenol both directly and indirectly. The company launched www.MedicineCabinetSafety.com, where visitors can get a free bottle of the OTC drug if they’re among the first 500,000 eligible people to register for one. For that portion of the campaign, which urges regular pruning of medicine cabinets for expired or recalled products and gives tips for their safe disposal, the company has recruited Suzy Cohen, RPh and author of The 24-Hour Pharmacist, to offer advice.
Those recalled products, of course, would include McNeil Consumer’s children’s and infants’ Tylenol and other OTC brands, subject to a cascading series of recalls due to manufacturing problems that started in April. As the Wall Street Journal noted, some of those products are going back on store shelves this week.
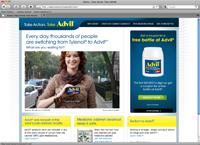
Visitors to www.takeactiontakeadvil.com can register to get free coupons there, as well, and can view a more blunt ad featuring upbeat music and the words: “Every day, thousands are switching from Tylenol to Advil. What are you waiting for?”
Pfizer, which took on Advil with its acquisition of Wyeth last year, is not the first competitor to kick Tylenol while it’s down. In July, Novartis announced plans to distribute 250,000 bottles of Triaminic Fever Reducer Pain Reliever, which competes with children’s Tylenol, free of charge as part of its launch of the line extension.